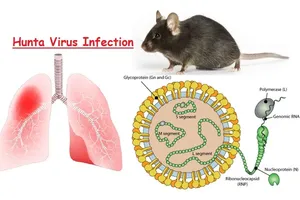
In a report submitted on May 6 to Prime Minister Le Minh Hung and Deputy Prime Minister Ho Quoc Dung, the ministry outlined the current disease situation and vaccine preparedness related to the SAT1 strain. FMD is a highly contagious transboundary disease affecting cloven-hoofed animals and is known for its rapid spread across regions and continents. The virus comprises seven serotypes: A, O, C, Asia1, SAT1, SAT2, and SAT3.
In Vietnam, only serotypes O, A, and Asia1 have been recorded in recent years, with no detection of SAT1 to date. Historically confined to Africa, the SAT1 strain has, since 2025, spread to several countries in the Middle East and West Asia. In early April, China reported its first SAT1 outbreak in cattle in Gansu and Xinjiang provinces.
According to the ministry, the rapid geographic expansion of SAT1 heightens the risk of its incursion into Vietnam, particularly amid complex cross-border trade and transport of animals and animal products. Should the virus enter the country, it could have severe repercussions for the livestock sector. Vietnam currently has approximately 31.4 million pigs, 6.14 million cattle, 1.95 million buffaloes, and over 3 million goats and sheep susceptible to FMD. In immunologically naive herds, infection rates could reach up to 100 percent without timely preventive measures.
The ministry noted that FMD vaccines currently licensed in Vietnam are effective only against serotypes O, A, and Asia1, offering no protection against SAT1. While SAT1 vaccines are available globally, they are not widely used in standard vaccination programs in Southeast Asia. Manufacturers include Biogenesis Bago (Argentina), DOLLVET (Turkey), Boehringer Ingelheim (France), Biopharma, KEVEVAPI (Kenya), and BVI (Botswana).
The ministry has received multiple requests from veterinary vaccine distributors and livestock producers seeking approval to import SAT1 vaccines for preventive immunization, with costs to be borne by farmers and enterprises.
Based on epidemiological evidence and existing legal provisions, the ministry has urged the government to permit the import of SAT1 vaccines already authorized for commercial use in other countries, to support emergency disease prevention efforts.
After quality inspections, imported vaccines would be authorized for use in livestock farms to establish proactive immunity and to implement ring vaccination in the event of an outbreak in Vietnam.