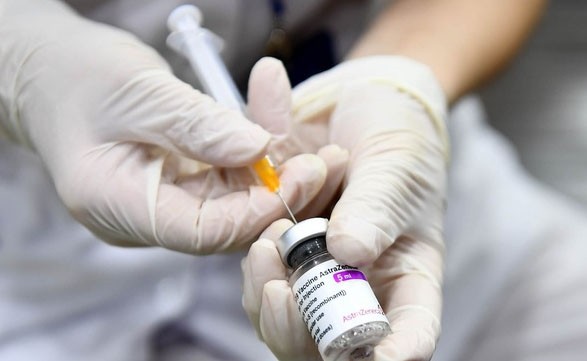
The information was provided on May 8 regarding the withdrawal of AstraZeneca Covid-19 vaccine globally from British pharmaceutical giant AstraZeneca.
The previously- licensed supply of AstraZeneca’s Covid-19 vaccine for circulation into Vietnam has expired and the country has not imported any doses of the vaccines so far.
At the current time, there are some other types of valid Covid-19 vaccines from other manufacturers being stored for use in the Covid-19 epidemic prevention and control.
A representative of the National Institute of Hygiene and Epidemiology said that AstraZeneca’s Covid-19 vaccine is a vaccine to protect people from the extremely serious risks of Covid-19 researched and developed by the British pharmaceutical company AstraZeneca in collaboration with the University of Oxford in the United Kingdom.
Additionally, it is also the first type of vaccine that the Ministry of Health conditionally approved for emergency use in the fight against the Covid-19 pandemic in Vietnam in February 2021.
At the period of being granted a circulation registration permit in Vietnam, AstraZeneca’s Covid-19 vaccine had been also granted licenses for emergency use in 181 countries and territories worldwide. Besides, the World Health Organization (WHO) also listed the vaccine in emergency usage.
In Vietnam, during the Covid-19 pandemic outbreak, apart from 30 million doses of the Covid-19 vaccine being ordered from AstraZeneca, the country partially received various batches of the vaccine through donation programs, therefore the vaccine had been used widely in the country for Covid-19 prevention and control.
Regarding the vaccination of AstraZeneca’s Covid-19 vaccine, Vietnam has safely run out of 74.3 million shots nationwide for adults aged 18 years and over. The last doses of AstraZeneca’s Covid-19 vaccine were used in advance of July 2023.
According to AstraZeneca, since May 7, the British pharmaceutical company’s Covid-19 vaccine has not been used anymore in the European Union as the company voluntarily withdrew its business license in this area.
Besides, AstraZeneca is performing the procedures of withdrawing licenses for the Covid-19 vaccine in other areas, regions and countries in the world.
The AstraZeneca Company explained that the vaccine recall was owing to commercial reasons as other vaccines are now more effective in the market against new SARS-CoV-2 variants, therefore the usage demand of AstraZeneca has been limited.
Recently, AstraZeneca’s Covid-19 vaccine was accused of having a link between the AstraZeneca vaccine and a rare side effect called thrombosis with thrombocytopenia syndrome.















)





