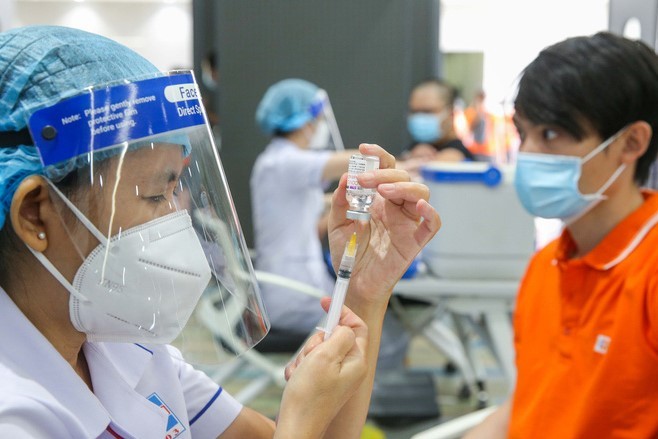
The press conference took place amid public concerns about the blood clot scares associated with the AstraZeneca Covid-19 vaccine as well as regarding the withdrawal of the vaccine from British pharmaceutical giant AstraZeneca and the proposal of terminating the usage of AstraZeneca Covid-19 vaccine globally.
According to the Ministry of Health, AstraZeneca vaccine is one among the 14 types of Covid-19 vaccines that the World Health Organization (WHO) issued its first emergency use validation on February 15, 2021 and the European Medicines Agency (EMA) approved to use in Europe from January 29, 2021.
Moreover, the vaccine was among the most populous Covid-19 vaccines in the world that issued emergency use validation in more than 170 countries with over two billion shots having been injected globally.
In reality, AstraZeneca vaccine was proven effective in preventing severe symptoms and reducing the death rate due to coronavirus. Database analytics from search works and the monitoring process after vaccination showed that the Covid-19 vaccine has been safe and effective for people of all ages.
Experiment results worldwide showed that vaccine effectiveness against SARS-CoV-2 infection reached 74 percent and there have not been any severe or critical cases being recorded with vaccinated people.
The WHO recommended that AstraZeneca Covid-19 prevention vaccine usage is safe and effective for adults aged 18 years or older.
Regarding a rare side effect called thrombosis with thrombocytopenia syndrome (TTS) due to AstraZeneca Covid-19 vaccine, statistics from the United Kingdom and Europe showed that one out of 100,000 adults had the risk of TTS within three days to 21 days after being vaccinated and only a few cases suffered from the side effect after 42 days of vaccination.
On the other hand, researches showed that the rate of AstraZeneca vaccine-induced immune thrombotic thrombocytopenia observed in cases after vaccinations was much lower than the rate in cases after Covid-19 infection.
Both WHO and EMA affirmed that the health benefits of vaccination with the AstraZeneca Covid-19 vaccine vastly outweigh the risks.
As for the usage of AstraZeneca vaccine against the Covid-19 pandemic outbreak in Vietnam, the Ministry of Health stated that the vaccine was granted a circulation registration permit nationwide in the fight against Covid-19 pandemic from February 1, 2021.
This is also the first Covid-19 vaccine that Vietnam imported for vaccination following the strict procedures and requirements of the Ministry of Health as well as roadmaps of the WHO and based on experiences in previous vaccination processes of advanced countries in the world.
Up to now, Vietnam has administered more than 266 million doses of Covid-19 vaccine to people aged five and older. Of which, 70 million doses of the AstraZeneca vaccine were used for initial and booster shots for individuals aged 18 and above.
Thanks to the widespread vaccination campaign with AstraZeneca Covid-19 vaccine together with other SARS-CoV-2 vaccines, Vietnam has well-controlled the spread of the epidemic and effectively reduced the infectious cases and death rate, contributing to bringing people back to normal lives.
The Ministry of Health of Vietnam recommended that a link between thrombosis with thrombocytopenia syndrome with the AstraZeneca Covid-19 vaccine is a rare side effect and only happens in a short period after being vaccinated.
From July of 2023, the country has run out of the AstraZeneca Covid-19 vaccine so the risk of AstraZeneca vaccine-induced immune thrombotic thrombocytopenia after being vaccinated with the British pharmaceutical company’s Covid-19 vaccine no longer exists.
Therefore, those who have been vaccinated with the AstraZeneca Covid-19 vaccine are not recommended to perform a D-dimer test or any test related to thrombosis with thrombocytopenia syndrome, emphasized the Ministry of Health.















)





