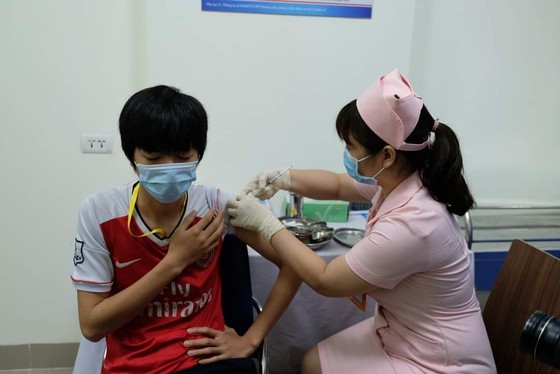
Six volunteers were given the shots of Covid-19 vaccine Covivac developed by the Institute of Vaccines and Medical Biologicals (IVAC). Speaking at the event, Deputy Health Minister Tran Van Thuan said he was delighted at witnessing the significant milestone in the development of Vietnam’s vaccine research and production.
He emphasized on strict procedures and trial techniques of vaccination as well as close post-vaccination observation to protect volunteers.
According to Professor Ta Van Thanh, Chairman of the science council of the school, said that researchers selected 120 healthy volunteers based on clinical tests. As scheduled, 120 volunteers will be divided into five groups; twenty of the 120 get a placebo while the rest will get shots of varying doses including 1 mcg, 3 mcg and 10 mcg.
In the morning, six first volunteers received placebo or vaccine and they were kept at the school in 24 hours for post-vaccination observation. Later, health workers will administer vaccine on 114 other volunteers at upcoming sessions, with 12-18 getting shots each time. The sessions will be spaced eight days apart and go on until April 20.
Eight days after immunization, volunteers must undergo health check-up and blood checks to evaluate the vaccine’s immunogenicity and the function of liver and kidney.
As scheduled, report of the first phase completion will be done in July, 2021. If the vaccine meets the safety standard and shows its good immunogenicity against Covid-19, the next trial will go along in the medical center in the Northern Province of Thai Binh.